Are Immune-Signaling Proteins The Key to Treating Alzheimer’s Disease?
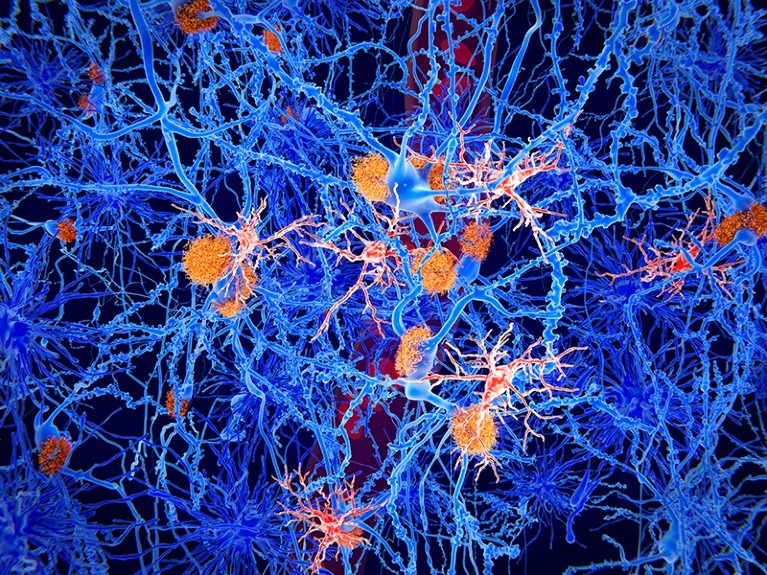
Above: Visualization of neurons with tau tangles represented in orange. Image courtesy of Rearden.
Alzheimer’s disease and related dementias place a substantial burden on families worldwide. An estimated 55 million individuals are currently living with dementia, with global costs surpassing $1.3 trillion dollars. Not only does dementia impact the individual, it also takes a massive toll on family members, care takers, and communities. Given the high disease burden and rapidly increasing prevalence in individuals 65 and older, recent scientific research has focused on understanding the mechanisms of the disease to inform the development of targeted therapies.
Current understanding of the mechanism underlying Alzheimer’s disease and other dementias suggests that an amyloid beta plaque interacts with tau proteins, located on the surface of neuronal dendritic spines. This causes the proteins to tangle and disrupt the structure of the dendrites, which are responsible for building connections, or synapses, between neurons. Eventually, these synapses are lost, disrupting the neuron’s ability to send signals to the rest of the brain that are important for memory retrieval and critical thinking. Furthermore, amyloid beta plaques and diseased tau proteins can propagate within the brain, spreading the dysfunction to other neurons. Ultimately, this leads to memory loss and cognitive impairment, hallmark features of Alzheimer’s and dementia.
However, scientists at the University of California San Francisco (UCSF) have identified cellular mechanisms that can break down pathogenic tau aggregates in the brain, reversing its effects on neurons. To do this, Drs. Kapmann and Samelson utilized a DNA screening technique called CRISPR interference (CRISPRi) which works by suppressing each gene in the neuron to see how it affects tau aggregation. They were able to identify multiple enzymes in the brain that modulate tau proteins. Among these, an immune signalling protein called CUL5 stood out as a promising candidate to protect a neuron from developing bundles of damaged tau.
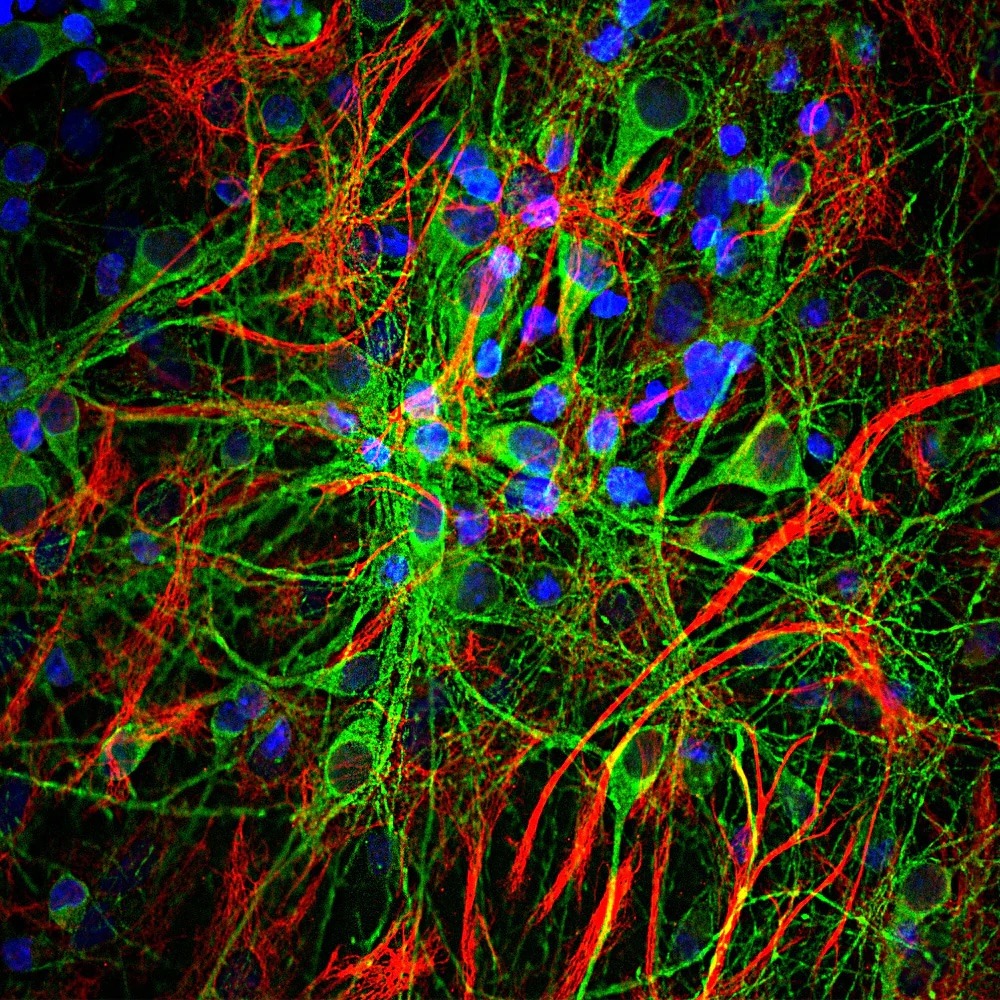
Above: Immunohistochemistry imaging of axons and glial cells. Neurons with tau are colored in green. Image Courtesy of EnCor Biotechnology Inc.
CUL5 functions by tagging and targeting proteins for cellular degradation. It also plays a role in inhibiting tumor growth in certain cancers, regulating viral and inflammatory immune responses, and neuronal development. Interestingly, an analysis of brain tissue from deceased Alzheimer’s patients showed that unaffected neurons had higher levels of CUL5. Therefore, Kapmann and Samelson believe that CUL5 increases a neuron’s resistance to accumulating damaged tau proteins by degrading the diseased tau proteins before they can form bundles.
An additional pathway related to tau aggregation involves the mitochondria, the organelle that produces cellular energy in the form of ATP. Certain genes impact mitochondria function, particularly in a process called oxidative phosphorylation where the majority of ATP is synthesized. Genes that decrease oxidative phosphorylation and disrupt cellular metabolism also increase the number of tau bundles in the neurons. Examining these genes and the effects of oxidative stress on tau proteins elucidates an alternative pathway in tau aggregation, suggesting that there are multiple molecular mechanisms impacting the pathophysiology of Alzheimer’s disease.
While the full mechanisms behind these two pathways are still unclear, identifying the role of CUL5 in tau degradation may have a profound impact on the future of Alzheimer’s research and other dementias. Future drugs may look to target CUL5 and modulate its expression.
