The Whale that Lives Two Lifetimes: What the Bowhead Whale Tells us About Cancer Resistance

Above: An aerial photo of a swimming bowhead whale. Image courtesy of NOAA Fisheries. Courtesy of Animals Around the Globe.
Human life expectancy has increased significantly over the course of history, increasing from 32 years in 1900 to between 70-80 years today. However, this pales in comparison to the estimated lifespan of the bowhead whale (Balaena mysticetus). As the second largest animal on earth, the bowhead whale has a lifespan of over 200 years! Remarkably, bowhead whales maintain low cancer development rates in spite of this long lifespan. In a study published in late October 2025, a group of scientists uncovered a mechanism for enhanced DNA repair in bowhead whales that may contribute to the whales’ exceptional lifespan and low cancer incidence. Not only do these findings provide insight into the factors affecting longevity and aging, but they may also have fascinating medical applications, especially in cancer treatment and prevention.
An Ocean Giant
The bowhead whale captured the attention of humans long before this study was published. As one of the few whale species exclusively found in Arctic and subarctic waters, the bowhead whale is well adapted for life in cold water, with extremely thick blubber and a distinctive arched upper jaw that helps them break through sea ice. Bowhead whales are classified as baleen whales, feeding mainly on krill and plankton. Like other whale species, bowhead whales form social groups that range from a few individuals to large aggregations that feed, migrate, and utilize a complex vocal repertoire to communicate.
Above: A group of swimming bowhead whales. Image courtesy of American Oceans.

From the 1800s to the early 1900s, bowhead whale populations were greatly depleted due to commercial whaling for their oil and baleen. Whalers targeted this species due to its slow swimming speeds and tendency to float after being killed. After commercial whaling was banned in the 1930s, the population has since recovered. Today, bowhead whales are listed in the Least Concern category by the IUCN Red List, though some populations remain classified as endangered.
Most background knowledge on the remarkable lifespans of bowhead whales came from observations from Indigenous communities. Native Alaskans and Canadians have been hunting bowhead whales for thousands of years, and the tradition is an incredibly important part of their culture. Through these hunts, native whalers have reported seeing the same whale over several generations. Captured whales have even been found with harpoon fragments that could be dated to over 100 years ago. Analysis of aspartic acid racemization from bowhead eye lenses also estimated some individuals to be over 100 years old, supporting the native whalers’ statement that bowheads have the lifespan of approximately “two human lifetimes.”
Above: Pangnirtung, an Inuit community in northern Canada, celebrates a successful bowhead whale hunt. Image courtesy of Nunatsiaq News.

These observations and findings have constructed a fascinating picture of the bowhead whale’s longevity. But what exactly is occurring at the molecular level that could be enabling their long lifespan?
Aging and Cancer from a Molecular Perspective
Aging and increased cell division heighten the chance of DNA mutation accumulation. Therefore, long lifespan and large size should theoretically increase predisposition to cancer. However, this doesn’t seem to be the case for all animals — many large organisms with long lifespans actually have lower observed cancer rates than smaller species. This phenomenon has been named Peto’s Paradox after epidemiologist Richard Peto, who described it in 1977.
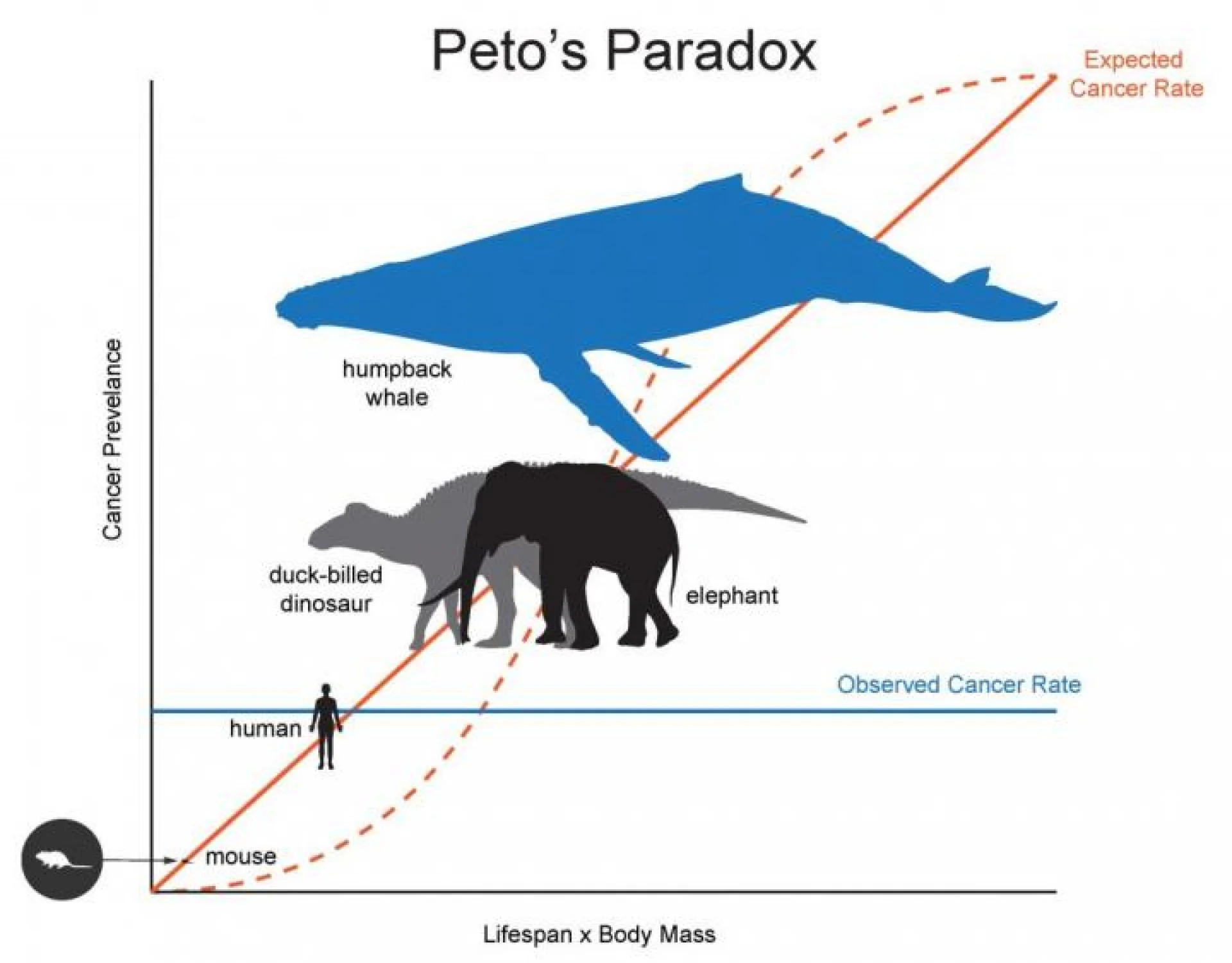
Above: A graphical illustration of Peto’s Paradox. Image courtesy of Interesting Engineering.
Peto’s Paradox has led scientists to study and compare these organisms with longer lifespans in search of identifying novel mechanisms of cancer avoidance. A well-known example occurs in elephants, which have a greater number of copies of tumor suppressor genes compared to humans. Researchers suggest these results are due to more robust elimination of mutated cells in elephants, as elephants have more copies of the tumor suppressor TP53 gene that triggers apoptosis in cells with DNA damage. Other comparative studies led to the development of the multi-stage model of carcinogenesis, which posits that a cell must undergo a certain number of genetic mutations for it to become a cancer cell. In this case, the cells of animals that are less prone to cancer require a greater number of mutations before they transform into cancer cells.
These examples demonstrate that mechanisms of cancer resistance vary widely across organisms. However, they do not significantly correlate with longer lifespans. What, then, could be behind the bowhead whale’s longevity and cancer resistance?
Identifying a Mechanism for Cancer Resistance and Longevity in the Bowhead Whale
To answer this question, the researchers — a large group affiliated with several institutions across the U.S. and Europe — first assayed bowhead whale cells for a number of mechanisms that could be involved in preventing cancer. These included comparing telomere length and telomerase activity, biomarkers associated with aging and longevity, and expression of tumor suppressor genes. Interestingly, bowhead whale cells did not exhibit significant differences in telomerase activity or tumor suppressor gene expression compared to human cells. In fact, they required fewer oncogenic mutations to transform into cancer cells! However, when exposed to radiation, whale cells displayed less apoptosis and expression of senescence-associated secretory phenotype (SASP) despite entering senescence, which may contribute to longevity.
Since cells required fewer oncogenic mutations to transform, the researchers hypothesized that cancer resistance may rely on enhanced DNA repair. In this study, the genomes of whale, human, and mouse-derived tumors were sequenced and compared to their parent non-transformed cells. Here, they found that bowhead whale tumors had decreased amounts of novel mutations than humans and mice when compared to their respective parent cells, as well as fewer numbers of structure-changing mutations. These results led the researchers to conclude that bowhead whale cells must be more resistant to accumulating mutations.
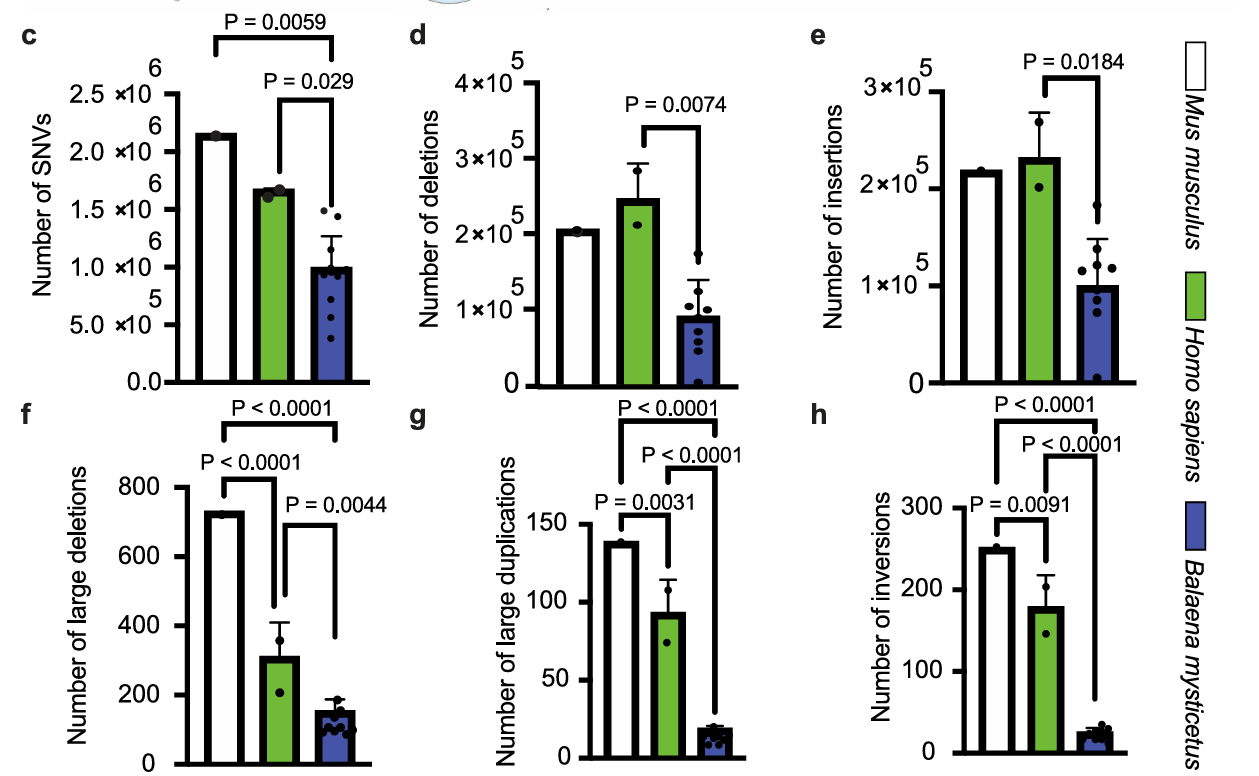
Above: Bar graphs quantifying different types of mutations in mouse (Mus musculus), human (Homo sapiens), and bowhead whale (Balaena mysticetus)-derived tumors. Small mutations (1-10 base pairs in size) are shown in c-e in the figure, while large mutations (>6 kilobases) are shown in f-h. SNVs, single nucleotide variants. Image courtesy of Firsanov et al. 2025.
How DNA Repair is Enhanced in Bowhead Whales
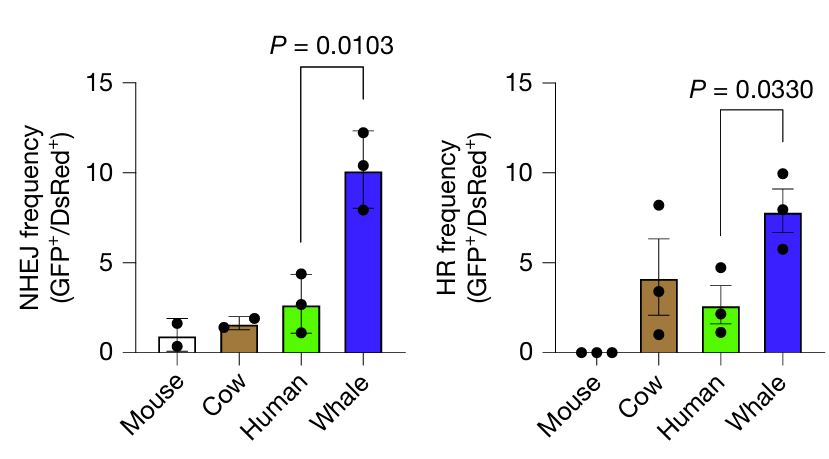
Next, the researchers sought to determine a mechanism by which bowhead whale cells prevented mutation. They assessed several DNA repair pathways and found that whale cells had higher activity in mismatch repair, particularly in double-strand break (DSB) repair, a pathway strongly associated with longevity. Both treating cells with damaging chemicals and using genome editing techniques to induce mutations showed that whale cells could resolve DSBs more rapidly and accurately than human cells.
Above: The frequency (left, middle) and accuracy (right) of DSB repair in humans, whales, mice, and cows. NHEJ, nonhomologous end joining. HR, homologous recombination. Image courtesy of Firsanov et al. 2025.
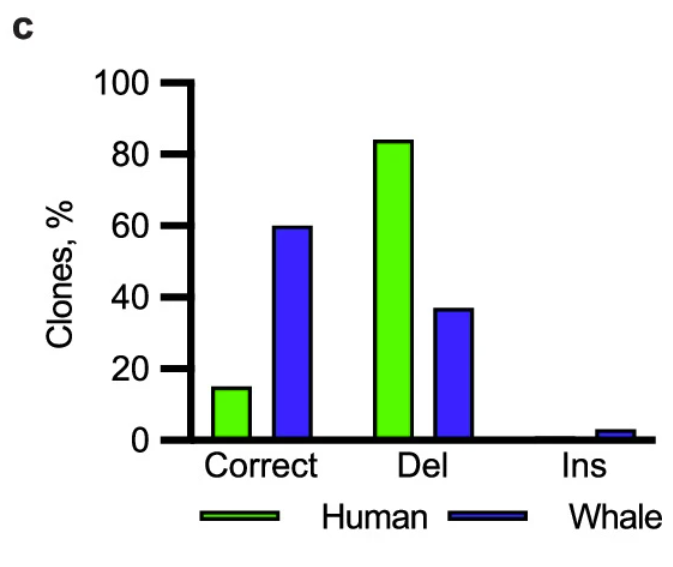
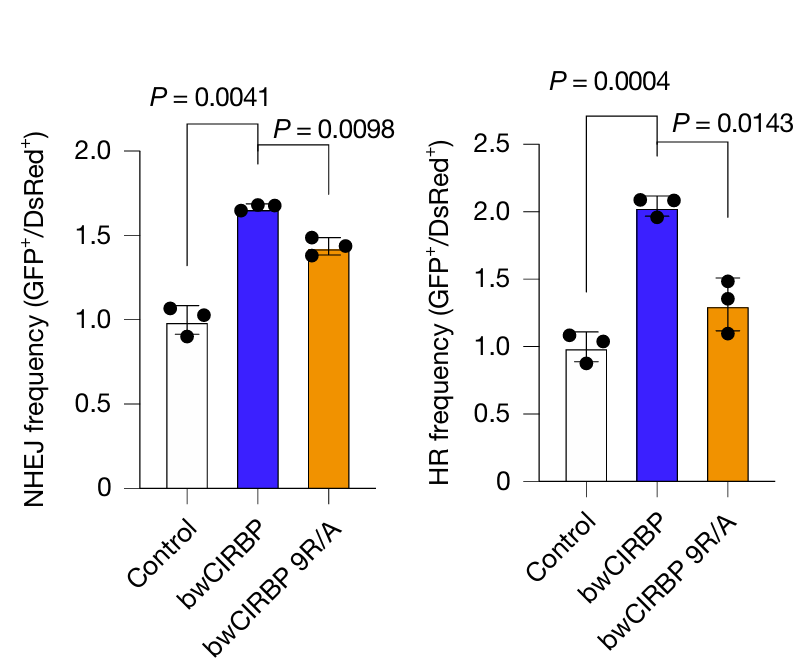
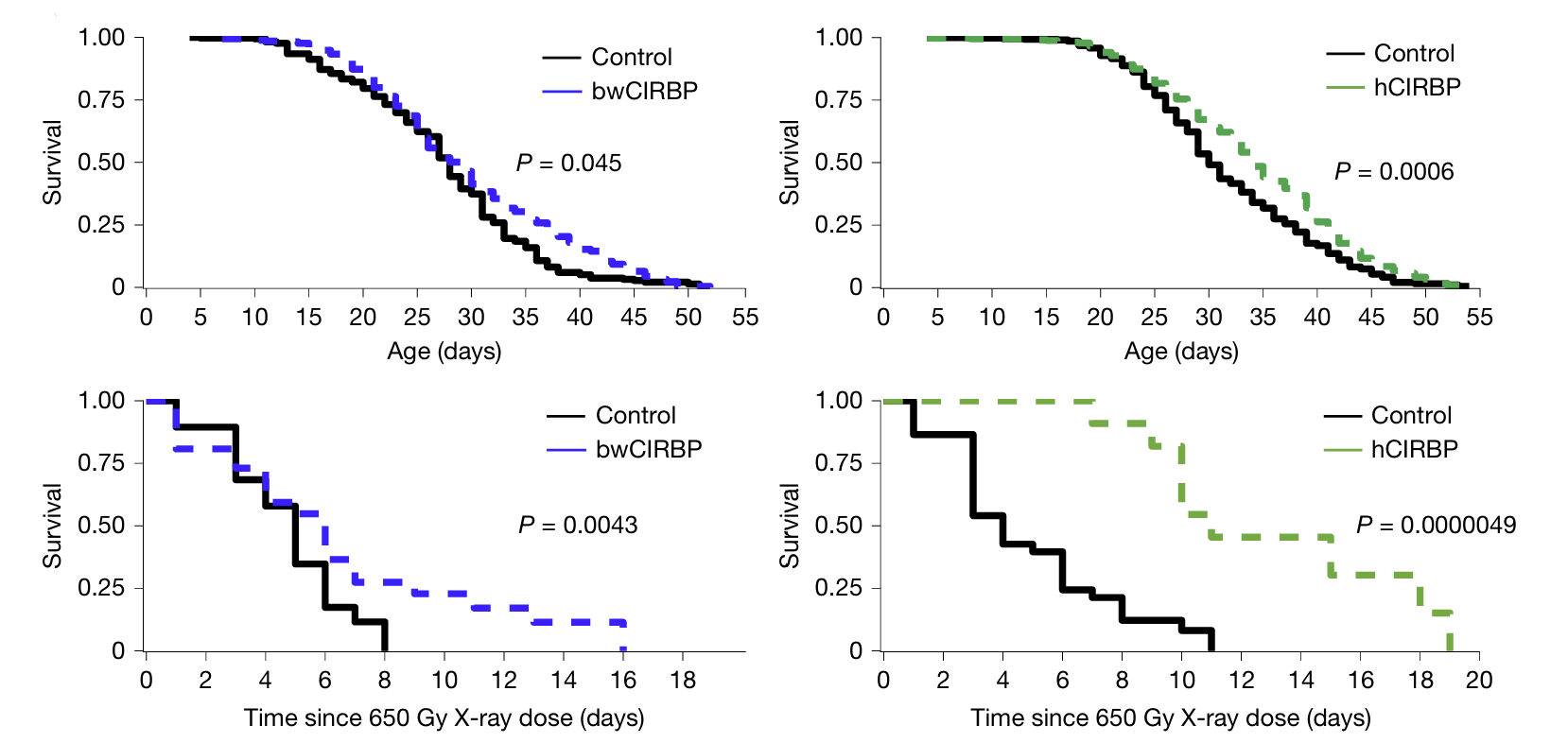
The researchers took this one step further by examining the expression of DNA repair-involved proteins across mammals using western blot. Here, they found that cold-inducible RNA-binding protein (CIRBP) was significantly abundant in bowhead whale tissues compared to other mammals. Substituting the bowhead whale CIRBP into human cells increased accurate DSB repair. Additionally, overexpression of CIRBP in fruit flies increased survival rates, especially after being exposed to radiation. Taken together, these results suggest that bowhead whale CIRBP promotes genome stability and longevity, contributing to this species’ remarkable cancer resistance.
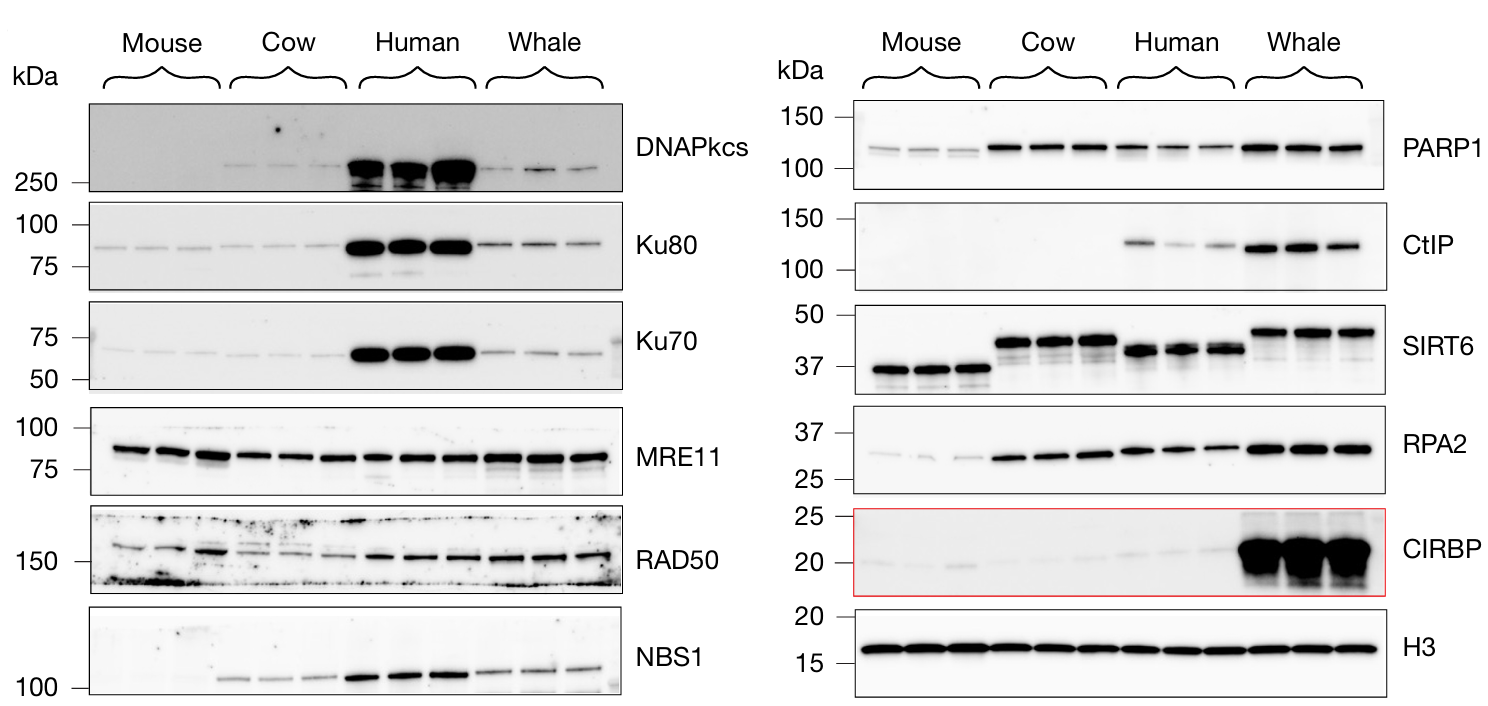
Above: Western blot showing levels of DNA repair proteins in bowhead whale cells (top), DSB repair frequency in bowhead whale CIRBP (bwCIRBP)-substituted human cells compared to those expressing mutated bwCIRBP (bwCIRBP 9R/A) (middle), and survival times of flies overexpressing either bwCIRBP or human CIRBP (hCIRBP) after exposure to X-rays (bottom). Images courtesy of Firsanov et al. 2025.
Looking to the Future
Though the findings with bowhead whale CIRBP strongly support its role in promoting cancer resistance, a few factors remain to be considered. The researchers state that the exact mechanism by which CIRBP enhances DSB repair remains to be determined. Furthermore, other proteins and pathways may also be involved. These unanswered questions warrant further investigation into this fascinating system.
These results raise the question of potential applications to cancer therapies. There are currently no approved treatments that involve DNA repair, and some suggest that this pathway may be impossible to improve. However, the researchers argue otherwise, pointing out how bowhead whale CIRBP enhances genome stability when expressed in human cells. Though designing safe, accessible treatments based on this system is likely still far off, these findings offer fascinating insights into the mechanisms underlying cancer resistance and longevity, and demonstrate the power of looking at the natural world around us for inspiration in improving human health and society.
