Where Cancers Hide: How Dormant Tumor Cells Evade Treatments and the Future of Prevention
Credit: Advanced Science News
The high prevalence of cancer recurrence in patients years after initial diagnosis, treatment and the “cancer free” declaration casts a shadow over the lives of survivors, forcing them to always keep this possibility in the back of their minds. For much of oncology’s history, the prevailing explanation for higher rates of cancer in previously diagnosed populations was straightforward: treatments failed to fully eradicate active disease. By this line of thinking, cancerous cells may continue to divide and spread because surgery may not remove all of a tumor, or chemotherapeutic treatments may not kill all of the cells. Surviving cancer cells would continue dividing at low levels, eventually becoming detectable again. However, several recent studies have proposed an alternate explanation for recurrence: cancer cells could enter a dormant, non-dividing state, hiding outside of the afflicted areas and surviving treatments, only to return to activity years or even decades later.
Researchers have found dormant tumor cells in a wide range of locations throughout the body, including bone marrow and lymph nodes. How and why these cells break away from tumors remains unknown, but they enter the bloodstream and circulate for a brief period before lodging in secondary sites and going dormant. With limited activity, chemotherapeutic treatments and other conventional methods of eliminating cancer cells tend to be ineffective on these cells.
The transition from small numbers to tumors resembling the complexity of the initial cancer has led many researchers to believe these dormant cells share the undifferentiated, adaptable qualities of stem cells. This stem-like flexibility could explain both why dormant cells resist elimination and how they reform complex tumors. Stem cells tend to stay dormant and respond in cases of injury or illness, a pattern that closely resembles these cancer cells and makes finding the triggers that re-activate them a top priority for current researchers.
Another particularly intriguing aspect of dormant cancer cells is their lack of feature retention in dormant states. Even though they produce new tumors that resemble those that had been previously treated, they lack consistent surface markers for reliable clinical detection, making genetic mutation tracking the only effective way to determine what kind of cancer a disseminated cell would reproduce.
A current hypothesis suggests that cells go dormant to survive immune-related attacks. Due to their separation from a larger and more insulated tumor, the cells become incredibly likely to be killed on their own when spreading through the bloodstream and to new sites. Studies have demonstrated that dormant cells alter genetic expression patterns along the mechanistic target of rapamycin (mTOR) pathway, which is key in regulating cellular metabolism and growth, in order to minimize their interactions with their surroundings and thus increase their chance of survival. In this limited state, dormant cells have been shown to gauge the state of the immune system and likely reactivate when changes create a vulnerable opportunity where proliferation is more survivable. Stimuli proposed as potential triggers in preclinical models include severe infections, COVID-19 and influenza, as well as general conditions like aging, severe stress and fibrosis.
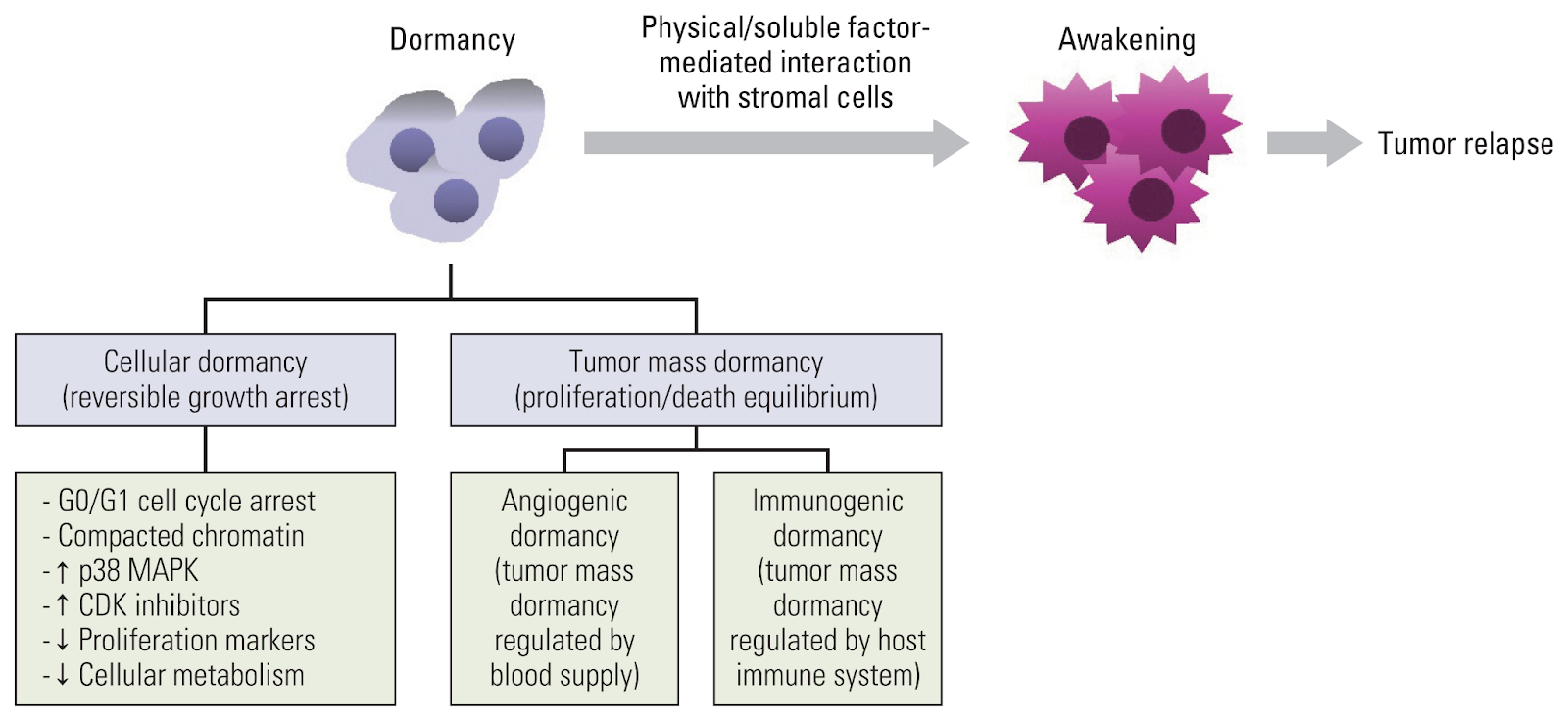
Above: A model demonstrating how dormant cells can awaken and cause tumor relapse. Image courtesy of Hye-Young Min and Ho-Young Lee, Cancer Research and Treatment.
Numerous research teams are now working to address dormancy and limit the development of secondary tumors by finding how to target and eliminate disseminated cells. Their efforts fall into two broad strategies: the first focusing on identifying and killing dormant cells, the second on making it extremely difficult for these cells to return to an active state. A team at the Albert Einstein College of Medicine has identified a potential pathway to target called the Integrated Stress Response Kinase PERK, which inhibits mTOR. By impeding this pathway with a drug called HC-5404 in animal models, the team disrupted the metabolic stability of dormant cells. Cancer biologists at the Fred Hutchinson Cancer Center in Seattle, Washington, are taking a different approach by focusing on revealing dormant cells to local immune cells, which they suggest may simply not encounter dormant cells frequently enough to eliminate them. Thus, they have worked on engineering CAR T cells — genetically altered immune cells from a patient — to increase immune activity and the likelihood of dormant cells being discovered and destroyed.
On the prevention side, several research teams have focused on controlling the stimulator of interferon gene (STING) pathway, a system by which the immune system detects cytosolic DNA from viruses, bacteria or damaged cells and prepares a defensive response. Researchers at Memorial Sloan Kettering Cancer Center are testing STING agonists to boost the efficacy of this pathway. They have found that STING signaling represses the progression of dormant cancer cells back into active ones. Additional research has shown that not only are the cells less likely to progress, but they also become far easier to eliminate by natural immune cells once these signals are upregulated.
While the ability of cancerous cells to hide away from the reach of therapies and the immune system is still a terrifying prospect for all cancer survivors, the multiple approaches being developed to eliminate these dormant cells are very promising. As research progresses and clinical trials develop, the presence of these cells in a significant minority of cancer survivors will drastically decrease, giving peace of mind and long-term security to patients.
