From Ponce de Leon’s 16th-century search for the Fountain of Youth to 45-year-old tech CEO Bryan Johnson investing millions every year to reverse his biological age, the process of aging has captivated humanity for centuries. At the cellular level, aging is defined as a gradual mechanistic decline that increases vulnerability to diseases such as cancer, neurodegeneration, and cardiovascular disorders. For years, the notion of manipulating age to expand the human lifespan seemed more like magic than science. However, rapid advancements in science – ranging from iPSC cells to epigenomics – have transformed that mystical ambition into a serious field of research.
To understand aging, it’s important to understand cellular differentiation, the idea that all of our cells have the same DNA, but vary widely in structure and function. Multicellular organisms begin as a single cell, yet develop into hundreds of distinct cell types – neurons, muscle cells, etc. This is done through selective gene expression, where the genetic landscape of each cell is sculpted by what we call epigenetic modifications (from the Greek root epi meaning “over, outside of, around”) caused by the environment. These modifications are vast, but many involve the addition of methyl groups to cytosine bases on DNA, blocking transcription into RNA, effectively stopping protein production in the cell. Others involve histone modifications, which change the way DNA is stored in space and thus can be accessed.
In 1957, Conrad Waddington related the concept of an epigenetic landscape to the cellular decision-making and differentiation during development, where the cell, represented by a ball, could take very specific and delineated trajectories, from which there was no way back. In this model, differentiation was deemed directional and decisions were irreversible, as was the common impression at the time.
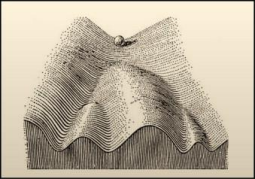
Above: An illustration of Waddington’s model of an epigenetic landscape. Photo courtesy of Goldberg et al.
However, in 2006, Shinya Yamanaka discovered four transcription factors that, when introduced to adult fibroblasts (primary cells of connective tissue), were able to wipe away these “epigenetic landscapes,” making them pluripotent stem cells, so that they could once again differentiate into many different types of cells. He won a Nobel Prize for this discovery and completely changed the landscape of biomedical science. Scientists could now take fibroblast cells from any patient and differentiate them into pluripotent stem cells, which could then be differentiated to another cell type for experimentation specific to that person's genome. Not only did this discovery add to the possibilities and expand the horizons of biotechnology, but it also changed the paradigm of thinking and expanded the possibilities, no longer limiting us to the Waddington model of cellular decision-making.
What does this have to do with aging? In a way, being able to change a cell from its adult form to its initial pluripotent form is a type of age reversal, showing that cells can travel backwards in their “epigenetic landscape.” Additionally, the epigenetic modifications that shape the cellular decision-making pathway are also involved in aging. For instance, in 1967, a study discovered that methylation patterns could be related to the age of spawning salmon. In 1987, it was found that the chromatin structure of the DNA in human skin fibroblasts exhibit changes with aging. And, in 2013, Dr. Steve Horvath proposed an epigenetic clock; he analyzed patterns of DNA methylation to correctly measure epigenetic age of a cell, measuring the cumulative effect of the epigenetic maintenance system and understanding aging across cell samples.
The existence of technologies like iPSC cells, along with knowledge of the role of epigenetic modifications in the aging process, has led to a new pursuit of epigenetic reprogramming to reverse aging and increase longevity. This perspective views aging as a disease, something that can be treated, rather than an inevitable process. Oftentimes, we have a symptoms-based approach to treating these age-related degenerative diseases and ignore the root problem: aging. Research of the aging cell is uncharted territory, where the unbelievable complexity of a single cell is combined with the billions of base pairs and epigenetic modifications – from DNA methylation to histone acetylation – that are all maintained dynamically, changing as we age. But to better treat degeneration and carcinogenesis, we must first understand how these systems maintain themselves.